Music: A Synthesis of Prenatal Stimuli
Robert Chuckrow, Ph.D.
©Copyright 1970, 2012 Robert Chuckrow
Note
This paper was first written during the 1960s but was not published (see pdf of scan of the original typewritten manuacript c. 1965: Music Paper (Original).pdf. What follows is an edited, updated version. Whereas the references are now quite dated, the content should still be valid. More recently (in 2017), Richard Parncutt and I published a paper entitled “Chuckrow’s theory of the prenatal origin of music” in Musicae Scientiae.
Introduction
For most people, listening to music is an experience accompanied by feelings ranging from mellowness, to excitement, to ecstasy. The question arises how an organized pattern of sounds can affect us so profoundly. Because the events we read or we see enacted in art forms such as literature or drama are directly connected to our own experience, we can usually understand how these art forms cause the feelings they do. In the case of music, however, the connection between the sounds we hear and our response to them is much harder to state. This difficulty suggests that music evokes responses formed during a very early period of life, during which conventional communication is absent.
This presentation will therefore consider what stimuli act on a fetus in utero and survey what is known concerning the fetal response to each type of stimulus. We will analyze these stimuli and establish a relationship between them and their effects on the fetus. Then we will consider the hypotheses that music is a form of communication beyond that of spoken language, which utilizes analogs of the main elements of prenatal stimuli. That is, just as spoken/written language has meaning to us in terms of our daily environment and experiences, the language of music has meaning to us in terms of our prenatal environment and experiences. We will then assert that inspired music speaks to and reminds us of our inner knowledge to use our creative abilities for bringing harmony into our world.
Factors in the Prenatal Environment
Taste, Smell, and Temperature
Under normal conditions, the fetus is surrounded by amniotic fluid of almost constant salinity, pH, and temperature. Thus, taste, smell, and temperature would not be expected to play an important role in the prenatal environment.
Tactile
Tactile stimuli are restricted to those caused by pressure fluctuations in the amniotic fluid, non-inertial forces (forces resulting from the mother’s changes in movement), and forces transmitted directly through the wall of the uterus. It is known that the quiescent fetus will actively respond to a directly transmitted force by squirming. Hooker1 reports that a fetal response to tactile stimuli can occur as early as eight weeks after fertilization of the egg.
Due to its density, the amniotic fluid does not exert uniform pressure. As the mother changes her position, the distribution of pressure on the fetus changes. The weight of the fetus (the force of gravity on the fetus), is always downward, and its direction with respect to the fetus therefore changes with the mother’s changes in position. Moreover, as the mother changes her state of motion, the fetus is moved by non-inertial forces in a manner similar to that of a passenger in a vehicle that accelerates or rounds a curve. Hiccups, a form of behavior commonly exhibited by fetuses, appears to be linked with a change in the mother’s position.2
Visual
In the ninth month of pregnancy the fetus’s eyelids cease to be fused. Even if the eyelids remain closed, we cannot assume that no light enters the eyes. Eyelids and even thicker layers of skin can transmit light. Whereas it is unlikely that the fetus receives any visual stimuli containing spatial patterns, there is a possibility that changes in the amount of light reach the fetal retina.
Acoustic
Present are the vibro-tactile and possibly electrical stimuli of the continual pulsing of both the maternal and fetal hearts. There are also the sounds of digestion and the vibrations produced by the circulation of the blood through the large arteries of the uterus. Also present are the regular sounds of the mother breathing and the intermittent sounds of the mother’s voice and other voices. There are sounds of chewing, swallowing, sneezing, coughing, yawning, brushing teeth, etc. Last, there are external sounds and noises such as that of clattering dishes, automobile horns, loud music, machines, etc.
The fetal response to loud sounds was noted as early as 1925 by Peiper3 and in 1927 by Forbes.4 Forbes mentions that a rap on the side of a tub in which a pregnant mother was bathing caused the fetus to jump within a fraction of a second. There is a question of whether the fetal response was tactile or auditory even though the mother was not conscious of feeling tactile sensations. Forbes also tells of a pregnant mother who was troubled by great fetal activity during periods of applause at a concert. Ray5 has confirmed that vibration will produce a fetal response. Sontag6 reports that an intense vibration coupled to the abdomen of a woman in her eighth month of pregnancy will occasion an immediate convulsive fetal response in nine out of ten cases. This response involves violent kicking and moving. In all cases there is an increase in fetal heart rate.
In addition, Bernard and Sontag7 have reported that the human fetus in utero is capable of perceiving a wide range of tones produced in the air, close to the abdomen of the mother. The fetus responds to tonal stimulation with sharp body movements and cardiac acceleration.
Oxygen, Nutrients, and Other Substances
Oxygen, sugar and other nutrients, and carbon dioxide are all in the prenatal environment, and their concentrations vary with the actions of the mother. Other substances such as stimulants, pharmaceuticals, and toxins may also be present and can cross the placenta, thus affecting the fetus.
Movement
Consider the stimuli to the fetus of a pregnant woman as she walks. Especially in the last months of pregnancy, walking produces a swaying and uneven movement of the fetus. Moreover, each step sends a thud-like vibration through her body, similar in sound and periodicity to that of the beat of a drum. Deeper breathing and consequent stepped-up oxygenation of the blood accompany walking and may well have an effect on the fetus.
Mothers know that cradling a baby in their arms and walking with a side-to-side rocking motion is very soothing to the baby.
Breathing
Shortly after the mother inhales, there is a consequent increase in oxygenation of her blood, and just before she exhales there is a corresponding decrease. The muscular action of breathing also has a massaging effect on the fetus. Each breath is accompanied by changes in pressure in the amniotic fluid. Of course, the air entering and exiting makes a recurring sound rich in high frequencies and is similar to the sound of cymbals in music. In popular music especially, the sound of cymbals “crashing” is very suggestive of the sound of the pregnant mother breathing. After the 2:30 point in the Beatles song Paul McCartney’s “Kreen-Akrore,” https://www.youtube.com/watch?v=eqSWPQMOg_Y, the repeated crash of cymbals is alternated with the sounds of inhalation and exhalation during actual breathing. Over the years, I have played this song in my lectures on the prenatal underpinnings of music.
Maternal Emotions
There is evidence that the emotions of the mother can affect the fetus. The experiencing of an emotion by the mother is accompanied by variations in the concentration of hormones, oxygen, glucose, and carbon dioxide in her blood. Further, it is known that all of these substances are capable of passing through the placenta to the fetus, which is thereby affected. The production by the fetus of hormones that have molecular sizes small enough to permit them to pass through the placenta is related to the rate of production of these hormones by the mother. That is, the endocrine systems of the mother and fetus do not act independently but interact to form a mutual pool. One example of this interaction occurs in the case of pregnant diabetic mothers. An infant born to a diabetic mother is prone to an overactive pancreas, which before birth, was stimulated to supply the additional insulin needs of the mother.8
At the time of writing this paper, little is known specifically of the immediate or long-range effects on the fetus of the mother’s emotions, although there is considerable evidence that the mother’s emotions do affect the fetus. Sontag9 links anxiety of the pregnant mother to a marked increase in fetal movement and to a greater incidence of irritability, hyperactivity, frequent stools, and feeding problems after birth.
Electromagnetic
At the time of writing this paper, the possible effects of variations in electric potential to which the fetus is subjected have not been investigated. This is an area which may prove to be important. For example, the rhythmic variations in the potentials associated with cardiovascular contractions may directly influence or even establish rhythmic variations of electric potentials in the fetal brain.
Environmental Patterns External to the Mother
Let us consider the existence of patterns in the fetal environment that might affect the fetus.
Speech. Speech conforms to rules of grammar and syntax and thus provides a source of acoustic stimuli containing meaningful patterns. Moreover, emotional states of the mother are not unrelated to patterns in her speech. Ostwald10 has attempted to analyze speech for such attributes as loudness and timbre and to relate these to mental states. Rubenstein11 found that tones hummed by subjects had a frequency pattern that varied diurnally. Thus, at the very least, diurnal changes in the mother’s voice would accompany changes in other factors that also vary diurnally and which affect the fetus.
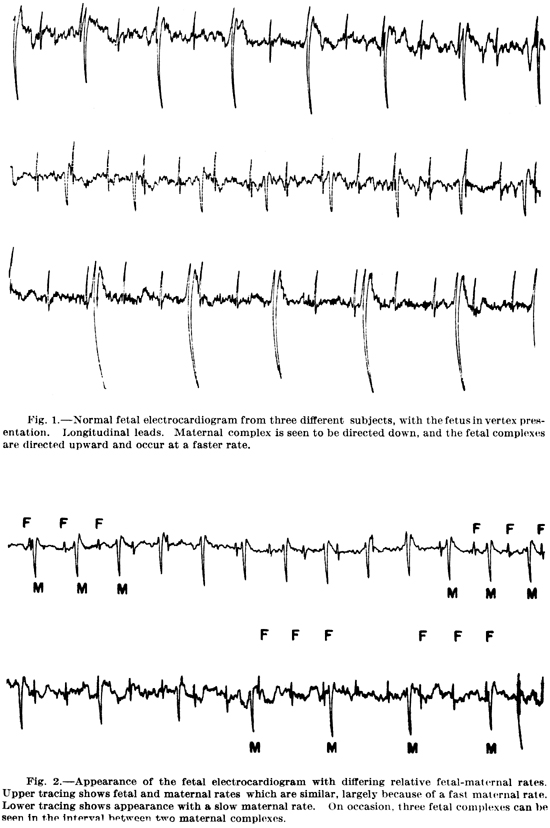
Heartbeat. Next, consider patterns formed by the combined pulsing of the hearts of the mother and fetus and the relation of such patterns to the emotional states and breathing of the mother. Contractions of the heart muscle of the fetus begin within a month following conception and become audible to the naked ear at the end of the fourth month. The fetal heart normally beats at a rate of 120 to 160 per minute, about twice that of the mother’s heart. The combined pulsations of the fetal and maternal hearts establish a definite rhythmical pattern which depends on the frequency and relative phase of the two pulsations (see Figs. 1 and 2). Any departure from the 2 to 1 ratio of fetal and maternal heart rates changes this pattern.
It is known that the heart rate of an adult varies with changes in physical activity, emotions, or breathing. Even in the absence of these changes, the heart rate of a normal individual alternately increases and decreases. This variation is termed the sinus arrhythmia. The increased rate usually occurs with respiratory inspiration, and the decreased rate occurs with expiration.
The fetal heart rate also undergoes a pattern of variation. These fetal variations are affected by external stimuli in the form of pressure, vibration, or chemical changes in the blood,12 but the pattern of variation appears to be independent of the maternal sinus arrhythmia.13
Thus in the absence of sudden changes in maternal emotions, activity, and breathing, and in the pertinent stimuli to the fetus, the combined pulsations of the hearts of the fetus and the mother produce a slowly varying rhythmic pattern (see Figs. 1 and 2). The sudden experiencing of an emotion by the mother would be accompanied by an immediate increase in her heart rate and the pressure of the pulsation. However, the fetus would not be immediately subjected to any chemical changes, and its heart rate would not be immediately affected. Thus, there would be an abrupt change in the combined rhythm (compare Fig. l to Fig. 2). This abrupt rhythm change would later be followed by chemical changes in the blood supplied to the fetus. The changes in rhythm are, therefore, a sensitive, early warning system, which enables the fetus to adjust to the chemical changes before they arrive, avoiding an unexpected jolt.
Fetal Learning
We next consider evidence that the fetus can learn from patterns in its environment. The most promising evidence that fetuses are capable of learning has been reported by Spelt.14 In an experiment with sixteen women in the last two months of pregnancy, Spelt conditioned the fetuses to respond to a vibro-tactile stimulus. Initially the vibro-tactile stimulus alone did not produce a response. Next, the fetuses were exposed simultaneously to both the vibro-tactile stimulus and a loud sound which, alone, did produce a response. After fifteen to twenty such paired stimulations, the fetuses were conditioned to the point at which three or four successive responses to the vibro-tactile stimulus alone could be expected. Additional conditioning raised the number of successive conditioned responses to as high as eleven.
Spelt’s experiment adds considerable plausibility to the idea that the fetus may learn responses to patterns inherent in prenatal stimuli. Greene15 suggests that oxygen, water, carbon dioxide, glucose, or their concentrations or combinations represent a kind of object for the fetus’s perception at one stage of development. Greene additionally proposes that vibrations, pulsations, and pressures incident to the mother’s physiologic activities, chiefly vascular and pulmonary, become an object during the later stages of fetal experience. Thus the consistent recurrence of these objects and the physiological and neuro-hormonal response of the fetus to them may constitute a sort of learning process. For example, during the last seven months of development, the fetus would be subjected to the order of 20,000,000 repetitions of alternating strong and weak cardiovascular pulsations. The minimum we can expect is that such stimuli leave a lasting imprint on the fetus. If so, the psychological phenomenon of grouping sounds would seem to have its roots in the cardiovascular elements of prenatal experience, as has been suggested by Lourie.16 Salk17 has provided experimental evidence that the cardiovascular prenatal stimuli do leave such an imprint. He has demonstrated that when newborn children are exposed to the amplified sound of a normal heartbeat, they are quieter, are healthier, and gain weight faster than when they are not so exposed. This work by Salk has been replicated by Tulloch et al.18
Specifically, let us attempt to picture the stimuli provided to the fetus by laughter on the part of the pregnant mother. Laughter, which involves contractions of the abdominal muscles and diaphragm, accompanied by high-pitched staccato vocal utterances, would constitute a definite climax to the pattern of speech sounds that evoked the laughter. These speech sounds would consist of phrases, repeated with gradual changes in words, intonation, and emphasis characteristic of humor. For the mother, the laughter would be accompanied by an exultant state and changes in her heart rate, breathing, and blood concentrations of oxygen and hormones. These changes would be expected to affect the fetus. The associated patterns involve a climactic change of acoustic, tactile, and chemical stimuli associated with a state of maternal well-being. If such patterns are repeated with sufficient frequency, they may become an object for fetal learning. According to this picture, the learning faculties of the fetus are not dormant until some definite moment when they suddenly become active but, instead, continuously grow. The difference between before and after birth may be more in the nature of the stimuli than in the degree to which learning may occur.
The Connection of Factors in the Prenatal Environment to Music
Let us next examine the idea that music utilizes analogs of the main elements of prenatal stimuli.
Beat, Meter, and Rhythm in Music
In almost all music there is a strong beat at a rate within a range approximately equal to that of the maternal heart rate. During a musical performance the tempo (the rate of the strong beat) is not constant but is varied in the context of the music in a manner analogous to the variation of the maternal heart rate with breathing, changing emotions, and physical activity. Moreover, in much music, this beat, which is produced by an instrument such as a drum or a plucked bass viol, has a sound resembling that of a pulsing heart. In addition to the strong beat, music contains weaker, but faster beats. The weaker beats are sensed by the listener as being grouped within the stronger ones. The particular pattern of grouping is called meter. For example, the meters that are formed by the main components of the combined maternal-fetal electrocardiograms in Fig. 2 are that of duple time (upper), triple time (lower first, second, third, and fifth groups), and quadruple time (lower fourth and sixth groups). The pattern determined by the time intervals separating the beats of a group (or measure) constitutes one type of rhythm. (Another type of rhythm is that established by the duration of tones.)
The following is a definition of rhythm from Webster’s Second International Unabridged Dictionary: “Music. a regularity or flow of movement which groups by recurrent heavy and light accent; accentual structure. Musical rhythm is based on the steady and persisting succession of beats or time units (pulses) within which accents periodically recur, these accents with their various degrees of stress serving to group the beats into measures. Measures in turn are subdivided into smaller groups that consititute the basis of form or design. ...”

Figs. 1 and 2. The above combined fetal and maternal electrocardiograms show the three basic patterns of maternal/fetal rhythms, namely one-to-one, one-to-two, and one-to-three. From S. D. Larks and K. Dasgupta, “Fetal Electrocardiography, with Special Reference to Early Pregnancy,” Am. Heart J., 56, 701–714 (1958).
Note that in each of the electrocardiograms shown in Figs. 1 and 2, above, the rhythm changes with time.
Most of the time the fetal heart rate is about twice the maternal heart rate, and the rhythm is slowly changing. A period of maternal emotional stress would be accompanied by a relatively large increase in rm and, consequently, correspondingly fast changes in rhythm. These changes would be accentuated by a greater intensity in the maternal pulsations.
If heart-rhythm patterns and the ensuing hormonal and chemical changes accompanying changes in maternal emotions constitute an object for fetal learning, then changes in maternal heart rate and, consequently, in rhythm would be expected to act as signals that precede chemical and hormonal changes. Thus, increases in maternal heart rate and fast changes in rhythm would be expected to produce in the fetus a state that is the prenatal analog of emotional tension. This tension would be further increased by an intensification of the maternal pulsation.
These mechanisms agree well with those by which music produces emotional tension formulated by composer/conductor, Howard Hanson.19 According to Hanson, an increase in tempo or changes in rhythm produce such tension in the listener, and the rhythmic effect is increased by an increase in dynamic intensity.
Actually more complicated rhythmic patterns are present in prenatal stimuli if we consider the weaker components of both the maternal and fetal complexes. These components would become more prominent under stress. Further, present knowledge does not establish whether or not the cardiovascular stimuli are most appropriately considered to be primarily acoustic, vibratory, or even possibly electric in nature. In any case, it appears that after birth, acoustic and vibratory stimuli can act as a satisfactory analog of the prenatal cardiovascular stimuli.
More on Beat
A few years after writing my original paper, I became aware of the idea that the “pounding” of the pregnant mother walking might be interconnected with music. I was sitting in my parked car in Manhattan, NY, listening to popular music, and watching people walk. I noticed that their footsteps closely matched the beat of the music that I, alone, was hearing. That coincidence suggested to me to think of how the footsteps of a pregnant woman would affect the fetus. I theorized that in some music—especially popular music—the main beat may not simulate the maternal heartbeat but, instead, the pounding of the mother walking. Many who listen to popular music like it to be so loud that they can actually feel the bass.
The feeling of walking is apparent in the music of the Bee Gees. When Discoursing to my students about the prenatal underpinnings of music, I would play various popular songs for them to illustrate my ideas. For one song, “Birdie Told Me,” by the Bee Gees https://www.youtube.com/watch?v=kihdhAqAeg4, I would walk back and forth across the front of the classroom, simulating the "pounding" of a woman in the late stages of pregnancy in sync with the beat of the song. More recently, one student pointed out that the movie, “Saturday Night Fever,” actually starts with people walking to a Bee Gees Song, "Night Fever" https://www.youtube.com/watch?v=u1qN6gLbUMw
,Many who listen to popular music like it to be so loud that they can actually feel the bass. I have tried walking with foam earplugs in my ear canals, which attenuates ambient sound and may somewhat damp the motion of my eardrums as amniotic fluid might for a fetus. In that mode, my feet contacting the ground sound very loud to me, and I can feel the effect throughout my body.
Tonal Patterns in Music
Music contains non-random tonal patterns vaguely resembling the rising and falling inflections and intonations and the syllabic articulations of human speech. Some music involves the singing of words, the presence of which suggests that music is closely related to human speech. It would seem that incorporating words ensures that melodic phrases conform to and exploit the structure of spoken language.
Feelings Evoked by Music
Some of the feelings evoked by music are a sense of buoyancy, a sensation of bathing in a warm fluid, an ecstatic swell of emotion or excitement, and an impulse to move in a manner related to the musical patterns, with a corresponding increase in pleasure when we obey this impulse. Considering these feelings in turn, the first two are highly descriptive of the womb. The ecstatic swell of emotion or excitement can be interpreted in terms of the conditioning influence on the fetus of maternal hormones associated with a state of well-being. We may picture the composer as one who possesses the ability to create patterns of sound, which when recognized by listeners, will produce in them a succession of feelings, the totality of which constitutes an experience they will desire to repeat. If these feelings result from learned responses to prenatal stimuli, then the composer is in touch with his or her own prenatal experience in a way that is shared by many others.
The impulse to dance or move to music may be explained as follows: Speech involves breathing and gesturing by the speaker in a manner related to the utterances. This breathing and gesturing is partly involuntary and partly an aid to expression for the speaker. The speech of a pregnant mother would therefore be accompanied by changes in pressure and force on the fetus, causing it to be moved in patterns related to that speech. Thus, supplying this motion ourselves intensifies the suggestion of the prenatal state.
The Ear and Harmony
If the ears of normal adults are subjected to a pure sinusoidal acoustic frequency f, as the intensity is increased they will successively begin to hear the additional frequencies 2f, 3f, 4f, ... . This phenomenon, called harmonic generation, is known to result from the inherent non linearity of the ear. If f is the frequency of the musical tone middle C (in this notation called c'), then the first ten terms in the harmonic series f, 2f, 3f, ... correspond to the musical tones c', c'', g'', c''', e''', g''', bflat''', c'''', d'''', and e'''', respectively (see Fig. 3). The octave, the interval f—2f (c'—c'') formed from the first two frequencies of the harmonic series, is found in the musical scale of every culture. The next two intervals, 2f—3f (c''—g'') and 3f—4f (g''—c'''), known as the fifth and fourth, respectively, are found in most primitive scales. We note that certain instruments such as the valveless trumpet naturally resonate at frequencies corresponding to a harmonic series, and the harmonics of a vibrating string (Fig. 3) likewise correspond to a harmonic series. If you click on https://www.historicaltuning.com/Harmonics.html, you will be able to hear the above harmonics separately and then together, as chords.
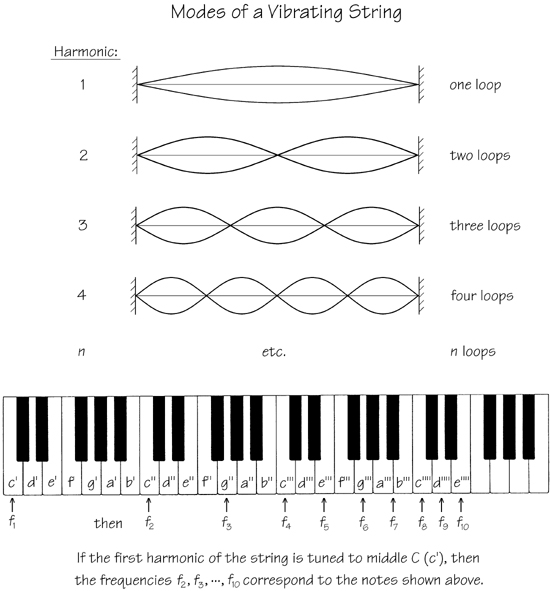
Fig. 3. Modes of vibration of a string fixed at both ends, along with their relative locations on the standard piano keyboard. Each loop represents the maximum displacement of the string above and below equilibrium (the vertical scale is exaggerated).
An interesting type of music is that of the Tibetan lamas. Intentionally incorporated in this music are sounds associated with bodily processes such as heartbeat, circulating blood, and even the ringing noise experienced during periods of silence. Tibetan music is designed to evoke, in the hearer and performer alike, a reproduction of what the lamas hear during meditation. In certain Tibetan monasteries, the lamas chant in a manner that results in the impression that each lama is singing a chord. Smith, Stevens, and Tomlinson21 have analyzed the spectrum of the voice of a single individual during this chanting, and they found that the fifth and tenth harmonics are highly accentuated. This incorporation of musical intervals in a single singing voice suggests the following hypothesis: The hearing of the fetus, although sufficiently developed to respond to acoustic stimuli, may involve more effects of harmonic generation than the hearing of an adult. Three possible mechanisms for harmonic generation seem worthy of consideration. (1) The fetal eardrum is contacted on both sides by amniotic fluid (rather than air for a neonatal), which may increase the inherent non-linearity of its vibration. (2) A greater non-linearity may result from incomplete development of structures in the fetal ear. (3) After birth the brain may learn to compensate for inherent distortion in the ear in such a manner as to raise thresholds for the subjective experience of hearing harmonics generated in the ear.
As a result of increased effects of harmonic generation, the fetus would “hear” a correspondingly greater proportion of higher harmonics than that for a neonatal subjected to a pure tone of the same loudness. Roughly speaking, for the fetus, pure tones may sound like chords. Thus, it appears that the inclusion of harmony in music may have its roots in prenatal stimuli.
Dissonance
Historically, music has shown a trend toward the inclusion of increasingly higher-order terms of the harmonic series, resulting in increasing dissonance. Bartholomew,21 in discussing this trend, states that “...it seems to be more than a historical coincidence that the order in which intervals have been accepted for use in music performed in parts parallels as closely as it does the order in which they occur in the harmonic series. The supposition is that, since most tones are complex, man’s ear had unconsciously been absorbing the sound of these intervals in the harmonic series of many of the tones he heard, and when he desired to use intervals for their own sake, they naturally appeared in musical history in the order of their appearance (and loudness) in the harmonic series.”
The increased effects of harmonic generation in fetal hearing mentioned earlier may be extended to account for the unconscious absorption suggested Bartholomew as follows: Consider the possible effects of music acting as a stimulus on successive generations of composers while in the womb. Each generation would be exposed to acoustic stimuli of a given degree of dissonance. This dissonance would be amplified by the presumed large effects of harmonic generation in the hearing of the fetus. When these fetuses grew up to be composers, they would naturally tend to make use of a higher degree of dissonance in their musical compositions. Thus, over the years, music would evolve in a direction of increasing dissonance.
It is tempting to apply this hypothesis to the question of why “too much” dissonance is disagreeable. In the womb, the highest degrees of dissonance are associated with the loudest sounds, and we know that loud sounds can produce a startle response in a fetus. Thus, highly dissonant sound, even if not loud, may evoke an echo of this response.
Formants and Counterpoint
Formants are distinct vocal frequencies that change from one syllable of a word to the next. It is known that a single human voice possesses a number of formants (Fig. 4). “Counterpoint is music consisting of two or more lines that sound simultaneously.”22 Note that in Fig. 4, there are at least four formants whose frequencies rise and fall as do the contrapuntal voices in the musical composition by J. S. Bach (Fig.5). Thus, the use of musical counterpoint may stem from prenatal exposure to formants in the maternal voice.
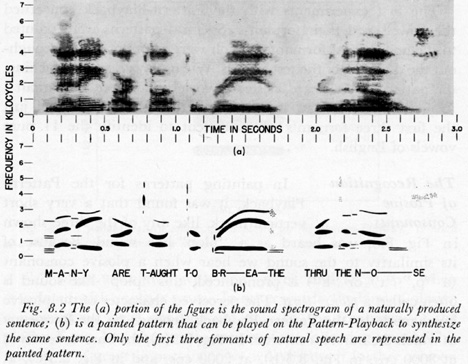
Fig. 4. Formants present in a typical person's voice. From Peter B. Denes and Elliot N. Pinson, The Speech Chain, Bell Telephone Laboratories, 1963, p. 129.

Fig. 5. Two lines of J.S. Bach's Minuet from the Third French Suite.
Summary
We have provided evidence that the fetus is exposed to, reacts to, and can learn from the vibrational, chemical, and physiological stimuli in its environment. The fetal stimuli include the mother’s voice, breathing, heartbeat, hormones, and body movement. Other stimuli are external sounds and the fetus’s own heartbeat. We have shown that these stimuli have analogues in music, namely harmony, phrasing, rhythm, counterpoint, and emotion.
Why is Music so Important to us?
Now that we have developed the idea that music utilizes patterns in the prenatal environment, we next ask what music generally communicates that is so important to us.
In the prenatal state, there is a recurring struggle to utilize hormonal and biological elements to balance and harmonize our environment. In that state, our responses mostly result from automatic, innate processes. After birth, however, we are thrust into a world wherein the struggle continues but in a different way. Now it is up to us to deal with the elements of our environment actively and consciously, and we are not always successful in transforming discord into harmony. When we are successful in doing so, we feel fulfilled and buoyed up; when we fail, we feel empty and frustrated.
Music involves a succession of abstract events, expressed in a language learned while in the fetal state. These events involve an exploration of disharmony and discord, which are then resolved harmoniously. The events in music are analogous to events encountered after birth, but now, success in resolving disharmony and discord is ingeniously built in by the composer. When we follow the events in music with our minds, we experience a taste of the joy that would be ours were we to apply our creative intelligence to harmoniously resolve the discordant events in our lives to the degree displayed in the music.
References
1Hooker, D., The Research Publications of the Association for Research in Nervous and Mental Diseases, 19, 237 (1939).
2Newberry, Norman E., “Fetal Hiccups,” J. Comp. Psych. 34, 65 (1941).
3Peiper, A., “Sinnesempfindungen des Kindes vor Seiner Geburt,” Monatsch. r. Kinderh., xxix, 236 (1925).
4Forbes, H. S. and Forbes, H. B., “Fetal Sense Reaction, Hearing,” J. Comp. Psychol., 7, 353 (1927).
5Ray, W. S., “A Preliminary Report of a Study of Fetal Conditioning,” Child Developm., 3, 175 (1932).
6Sontag, L. W., “Implications of Fetal Behavior and Environment for Adult Personalities,” Ann. N. Y. Acad. Sci., 134, 782 (1966).
7Bernard, J. and Sontag, L. W., “Fetal Reactivity to Tonal Stimulation, A Preliminary Report,” J. Genetic Psych., 70, 205 (1947).
8Gordon, Ohio State Med. J., 32, 540 (1936).
9Sontag, L. W., “The Significance of Fetal Environmental Differences,” Am. J. Obstet. & Gynec., 42, 996 (1941).
10Ostwald, P. F., “Acoustic Methods in Psychiatry,” Scientific Am., 212, 82, (Mar 1965).
11Rubenstein, L., “Humming, A Vocal Standard With a Diurnal Variation,” Science, 134, 1519, Nov. (1961).
12Sontag, L. W. and Newberry, H., “Incidence and Nature of Fetal Arrhythmias,” Am.J. Dis. Child., 62, 991 (1941).
13Sontag, L. W., “Individual Differences in Heart Rate Variability in the Human Fetus,” Am. J. Obstet. & Gynec., 98, 61 (1967).
14Spelt, D. K., “The Conditioning of the Human Fetus in Utero,” J. ExPer. Psychol., 38, 338 (1948).
15Greene, W. A., Jr., “Early Object Relations, Somatic, and Personal,” J. Nerv. & Ment. Dis., 126, 225 (1958).
16Lourie, R. S., “The Role of Rhythmic Patterns in Childhood,” Am. J. Psychiat., 105, 653 (1949).
17Salk, L., “Effects Or the Normal Heartbeat Sound on the Behavior of the Newly-Born Infant, Implications for mental Health,” World Mental Health., 12, 168 (1960)
18Tulloch, J. D., Brown, B. S., Jacobs, H. L., Prugh, D. G., and Greene, W. A., “Normal Heartbeat Sound and the Behavior of Newborn Infants—A Replication Study,” Psychosomatic Med., 26, 661 (1964).
19Hanson, H., “Some Objective Studies of Rhythm in Music,” Am. J. Psychiat., 101, 364 (1944).
20Smith, H., Stevens, K. N., and Tomlinson, R. S., “On an Unusual Mode of Chanting by Certain Tibetan Lamas,” J. Acoustic Soc. Am., 41, 1262 (1967).
21Bartholomew, W. T., Acoustics of Music, (Prentice-Hall, Inc., Englewood Cliffs, New Jersey, 1942), pp. 164-5.
22Willi Apel, Harvard Dictionary of Music, Belknap Press of Harvard University Press, Cambridge, MA, 1969, p. 208
Links to Interesting Articles on the Internet
http://superconscious1.blogspot.com/2008/01/prenatal-learning.html
©Copyright 2012 by Robert Chuckrow